Disaster Victim Identification
Due to New York State Department of Health regulations, we cannot offer any DNA tests to New York residents. Click here if require more info about DNA testing in NY.
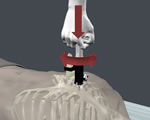
The DVIstat™ tissue extraction device is a practical tool for securing tissue samples from cadavers in mass fatality incidents while simultaneously securing an active Radio-Frequency Identification Tag (RFID) to facilitate electronic control of all subsequent movements and location in a mass interment situation.
 |
 |
 |
 |
| 1 | 2 | 3 | 4 |
 |
 |
 |
 |
| 5 | 6 | 7 | 8 |
SADE ID (Safe Accurate DNA Extraction Devices) is licensed worldwide to manufacture and sell DVIstat™ device as the preferred method of extracting core samples for DNA analysis and tracking of the victims until they can be returned to their families.
Please see below for a copy of the PowerPoint presentation that outlines the benefits and applications of the DVIstat™ device – click to activate, then use the Page Up/Down keys (Firefox only) or click to move between slides.
We envision each DVI team will stock a relevant quantity of devices to match their obligation under their countries national disaster plan. In addition, we will contract to hold in stock an agreeable quantity of additional devices to be ready for deployment, within an acceptable timeframe, to said DVI team should they call for them (known as ‘Off the Shelf’).A demonstration video of the DVIstat™ device is available upon request. Please contact us by email at [email protected]
DVIstat™ tissue extraction device exists to make your job easier, reduce or eliminate errors, hasten the identification process and, not inconsequentially, to drastically reduce the costs currently borne to establish identity of a victim via DNA analysis.
To facilitate this there are a number of key elements that must be understood:
Sample DNA analysis is NOT included in the price of the DVI tagging device. Shelf Life – the batteries within the RFID device have a shelf life of 7 years, these would need to be renewed after 5 years. Each RFID would be checked at every annual service and replaced for a charge if found to be inadequate. Preservation solution would need to be replaced at 5 years.
‘Off the Shelf’ contract – additional devices would be available from us, under contract, furnished to the registered offices of the DVI team, within an agreed upon number of calendar days of request. The quantity of additional devices would be an agreed upon quantity. For example: double that held by the DVI team (i.e. if 1,000 units are purchased, we would have an additional 2,000 available to them).
The cost of this service is paid annually in advance and is a percentage of the purchase cost. We would be responsible for maintaining the ‘Off the shelf’ units and the customer would be responsible for delivery costs.
Annual service and training to be contracted at agreed daily rate.
If you would like to discuss the DVIstat™ device further, please contact us by email at [email protected]



